 
High-throughput sequencing approaches have been commercially available for over a decade, and the technologies continue to evolve and improve ( 13). 
NGS is a term used to represent different technologies that enable massively parallel sequencing of clonally amplified or single DNA molecules. This review highlights currently available standards and guidelines for the implementation of NGS in the clinical and public health laboratory setting, and it includes considerations for NGS test validation, QC procedures, PT, and reference materials. 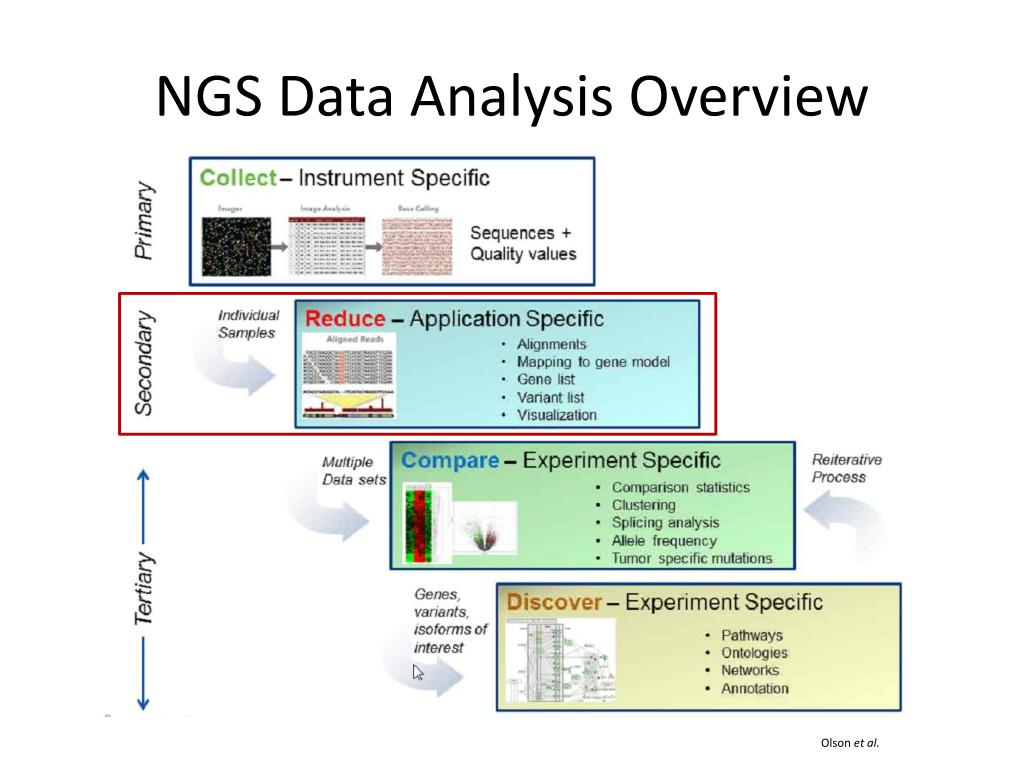
These include reducing the cost and turnaround time of sequencing, the development of fully automated user-friendly sequencing and data analysis pipelines, the creation of comprehensive and well-curated reference genome databases, curation of genotype-phenotype correlations for clinically relevant microorganisms (for example, when making predictions about antimicrobial resistance), establishment of proficiency testing (PT) and quality control (QC) measures, and the development of practice guidelines to ensure the quality of NGS-based tests ( 7, 9, – 12). Additional issues have been identified that should be addressed to fully realize the integration of NGS into the clinical and public health laboratory setting. 
Several issues and capacity gaps were identified by the 2014 APHL survey, including the need to identify public health laboratory NGS applications beyond sequencing of foodborne pathogens, the development and support of information technology (IT) infrastructure, and the need for training of public health laboratorians ( 7). For example, the New York State Department of Health is now using whole-genome sequencing to track the emergence of drug resistance for influenza virus ( 8). The APHL survey also indicated that the use of NGS in public health laboratories is expected to expand and be applied to an increasing diversity of public health investigations and applications ( 7). A 2014 survey conducted by the Association of Public Health Laboratories (APHL) revealed that public health laboratories are embracing the adoption of NGS technologies, especially for foodborne pathogen surveillance activities ( 7). Both the emergence of affordable and user-friendly benchtop sequencers and the resources and funding made available through federal initiatives have helped transition NGS into public health laboratories. Department of Agriculture/Food Safety and Inspection Service (USDA/FSIS) have established an Interagency Collaboration on Genomics and Food Safety (Gen-FS), with the goal of fostering timely access to genomic data for foodborne pathogen surveillance and outbreak response ( ). The CDC, Food and Drug Administration (FDA), National Institutes of Health (NIH), National Center for Biotechnology Information (NCBI), National Library of Medicine, and the U.S. AMD initiatives include broad applications of NGS to address public health problems, including vaccine improvement, identification of emerging threats, and tracking diseases and outbreaks ( ). Centers for Disease Control and Prevention (CDC) Advanced Molecular Detection (AMD) Initiative, national, state, and local partners are beginning to incorporate NGS-based methods into disease surveillance systems. A number of agencies are working to bring NGS into the public health laboratory setting. For example, the use of metagenomics combined with a rapid bioinformatics pipeline recently facilitated a clinically actionable diagnosis of neuroleptospirosis when conventional testing was initially unable to identify the causative organism ( 6). Metagenomic “unbiased” NGS applications, coupled with recently developed bioinformatics solutions ( 3, – 5) that enable the identification of all pathogens directly from a clinical sample based on sequence homology, have the potential to complement or even replace current standard clinical laboratory tests. For example, NGS has recently been used to better understand hospital outbreaks and inform infection control practices ( 1), and it can be used in the clinical microbiology laboratory to identify unknown organisms, predict antimicrobial resistance, assess virulence gene content, and inform molecular epidemiology efforts ( 2). The applications of NGS are wide-ranging and include whole-genome sequencing, microbiome analysis/metagenomics, transcriptome profiling, infectious disease diagnosis, pathogen discovery, and public health surveillance. Next-generation sequencing (NGS) is transforming the landscape of clinical microbiology and public health laboratories. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
