 So now that we know the formal charge formula, click on to the next video on, Let's put it to practice with the given example. This is the sum off all formal charges within a compound. We're going to say remember Valence electrons equals the group number off that element, and when we're talking about non bonding electrons, realized that we're gonna count them individually with formal charge when we find the formal charge of every element within a compound, we can calculate what's called the Net charge. We'd say formal charge equals the valence electrons off that element that we're examining minus the bonds that it is making, plus the non bonding electrons that it possesses. Bonding electrons are the electrons that do participate in bonding with other elements, and non bonding electrons are the electrons that don't participate and bonding with other elements with formal charge. (1) F C V N B 2 where V is the number of valence electrons of the neutral atom in isolation (in its ground state) N is the number of non-bonding valence electrons on this atom in the molecule and B is the total number of electrons shared in bonds with other atoms in the molecule. 
We assume that all atoms that are sharing electrons between each other they share them equally now with formal charge were introduced to terms such as bonding, electrons versus non bonding electrons. But when it comes to formal charge, we don't take that into consideration. We can double-check formal charge calculations by determining the sum of the formal charges for the whole structure. In reality, sometimes an element that's more electro negative will hold on to that electron within the electrons within the bond a little bit more tightly around themselves, which inflates or makes the electron clouds larger. Thus, we calculate formal charge as follows: (4.3.1) formal charge valence shell electrons (free atom) lone pair electrons 1 2 bonding electrons. The geometry of Molecules is a platform where we try to educate you about various chemistry concepts and find the answers you have been looking for.Now formal charge represents the charge given toe elements when assuming electrons are shared equally, regardless off electoral negativity. And it might seem challenging, but it becomes the most fun and easy subject to learn if learned the correct way. Sounds easy, right? Go ahead and try our formal charge calculator.ĭo you know that a perfect diamond is made up of a single Carbon molecule? Chemistry is one of the most exciting subjects. And to make it easy for you to do the calculations we have developed a Formal charge calculator for you, where you have to just enter the number of valence electrons, non-bonding electrons, and bonding electrons to find out the formal charge. 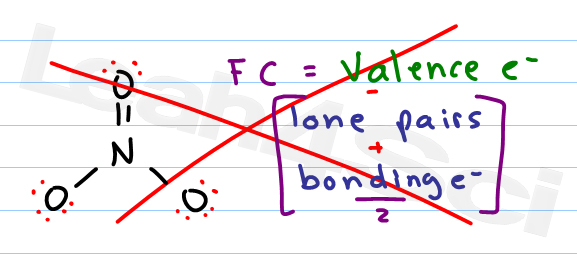
Similarly, you can use this formula for other molecules and find out formal charges for individual atoms in the molecule. And using the formula stated above, we found out that the total charges on both Sulphur and Fluorine atoms are zero. Here in SF4 as all the Fluorine atoms are arranged symmetrically, they will have the same charges. 
of valence electrons Non-bonding electrons Bonding electrons Total charge Sulphur (S) Now that we know the formula let us look at the example of how to find out formal charges for individual atoms in a polyatomic molecule Let us calculate the formal charges of SF4 Atom Total no. One can calculate the formal charges for any given atom with the help of the following formula:į.C = Valence electrons – Nonbonding electrons- Bonding electrons/2 
These charges help in knowing if the given structure of the molecule is stable or not. Formal charge is the individual electric charges on the atoms in a given polyatomic molecule. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
