 
With the use of Carbonic Acid through decomposition.Ĭarbon Dioxide also known as CO2 has a molar mass of 44.009 g/mol with a density of 1562 Kg/m3.\): Geometric structure of methyl isocyanate (CH 3–N=C=O), note there is no rotation around the double bonds only the single CN bond can rotate. For understanding the concept of the molecular geometry of co2 we need the Lewis structure of carbon dioxide. Because oxygen atoms make sigma bonds with the central carbon atom to complete their octet, CO2 has a linear molecule shape. Molecular Geometry and Bond Angle of CO2. Calcium Carbonate goes through the heating process and forms quicklime which is commonly used in industries.Ĥ. In CO2 molecular geometry, carbon makes a double bond with each of the two oxygen atoms, resulting in a tiny symmetrical, linear molecule of CO2 that is volatile and reasonably reactive. This process uses combustion, where carbon-based fuels are produced through thermal decomposition. This is where calcium carbonate reacts with hydrochloric acid, which is a very common method used in laboratories.ģ. The carbon-oxygen ratio in a CO 2 molecule is 1:2. 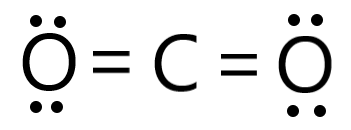
The H 2 S molecule possesses a mutual bond angle of 92.1° between the bonded atoms. Carbon dioxide is a colourless, odourless, incombustible gas produced by the combustion of carbon. H 2 S does not possess any hybridization as per Drago’s rule. The ideal electron pair geometry of H 2 S is tetrahedral. There are various processes in preparing CO 2.ġ. The molecular geometry or shape of H 2 S is bent, angular or V-shaped. SO2 Lewis Structure Preparation Process The molecule of chloroform (with tetrahedral shape CHCl3 molecular geometry) is tilted at 109.5 degrees bond angle of Cl-C-H. Which of these molecule(s) is polar Why is there a difference in bonding angles between water and carbon dioxide CO2.Carbon Dioxide (1) has two double bonds, which contribute to its linear geometry. This is due to the different numbers of electrons in each molecule and VSEPR (Valence Shell. There is no dipole moment generated between the two oxygen atoms making this not a polar molecule. Why is CO2 a linear molecule whereas H2O has a v-shaped geometry. When one or more of the bonding pairs of electrons is replaced with a lone pair, the molecular geometry (actual. The total number of electron pairs, both bonding pairs and lone pairs, leads to what is called the electron domain geometry. The CO2 molecular geometry is linear in shape, which means that the double bond has the same influence on the charge, making CO 2 a nonpolar molecule. The molecular geometries of molecules change when the central atom has one or more lone pairs of electrons. There are eighth electron pairs in this bond and no lone pair as shown in the Lewis Structures. This means that CO2 molecules are non-polar and will not be very soluble in water (a polar solvent). For example, carbon dioxide is a linear molecule. Explanation: The shape of a molecule helps to determine its properties. Two oxygen atoms are at the terminal ends of the bond, while the central carbon atom has an angle bond length of 180°. Answer: Molecular geometry is used to determine the shapes of molecules. CO 2 has double bonds, coming from Carbon and Oxygen. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
